How many times have you tried a new product only to find out that it didn't work for your skin,...
How To Buy Skincare Online
At 3AM I was doing my Instagram duty – checking Explore and looking for the next big trend when a promoted skincare post interrupted my scroll. It was gorgeous. The model was showing off the smooth serum as she applied it to her skin – the background was bleached out by a filter, and there was just enough fluffy things with hints of pink and gold that made me utter an audible “I want”.
But what was up with this skincare? I had never heard of it before, in fact most skincare that hits my feed lately seems to be new. Is it white-label, private-label, a new spin-off brand? I had no idea, but the ad was effective so I was going to find out.
When it comes to buying skincare online there are important questions to ask. In this post we’re going to explore how to read the label of a skincare product, understanding ingredients, and qualifying the product for use and sale in Canada.
Reading The Label - What's In Your Product?
Whether its food or skincare your ingredients lists works the same way. The ingredients listed on the label are listed in descending order of predominance, in their concentration by weight. The first item listed will have the most dominant concentration, and the last ingredient on the list will be the least.
Seriously, What's Really In Your Product?
In order for a skincare or cosmetic product to be qualified for use and sale in Canada the ingredients and their concentrations (or concentration ranges) have to be declared to The Consumer Safety Directorate at Health Canada using the Cosmetic Notification Process. Health Canada warns that this Notification Process IS NOT an approval process, it’s just an administrative process. So while Health Canada does not approve or disapprove cosmetics they will advise the filer if something seems wrong and not issue a CNF number.
The individual ingredients on the label should be listed by their INCI name. Curious about the safety and function of these individual ingredients? Look them up in the INCIDecoder. This helpful database will identify what these ingredients do, and how safe they are.
Under the regulations in Canada all cosmetics must also:
Have labels that are in both English and French, and be clearly legible for the life of the product.
There are many more interesting and very specific requirements for cosmetics and cosmetic labelling in Canada that are worth reading.
Be sure to also check out the Cosmetic Ingredient Hotlist. This list contains important restricted items, or restricted concentrations of ingredients and is published by Health Canada.
The Problem With Cosmetic Labelling and Regulation in Canada
Any product deemed to be a drug, either because it claims to have therapeutic properties in its marketing or in fact has therapeutic effect will have a DIN (Drug Identification Number) assigned to it. This DIN must be displayed on the label. Therapeutic product without a DIN? Potentially ineffective, or unsafe.
But what about other cosmetics that have been registered with Health Canada? How can you tell? You cannot. The registration numbers for cosmetics are not to be displayed on the cosmetics themselves and this creates a problem for the consumer.
It gets worse. A manufacturer that has qualified its product for use and sale in Canada isn’t technically allowed to advertise that they have done so. This rule is baked into the Cosmetic Regulations C.R.C., c. 869 which says: “No reference, direct or indirect, to the Act or these Regulations shall be made on any label or in any advertisement of a cosmetic unless the reference is a specific requirement of the Act or these Regulations.”
So if it is safe then there is only limited language that can be used to tell consumers that it is.
So anyone labelling “Health Canada Approved” on any of their cosmetics is operating under a big “no-no”.
Signs That Your Cosmetic or Skincare May Be Unsafe
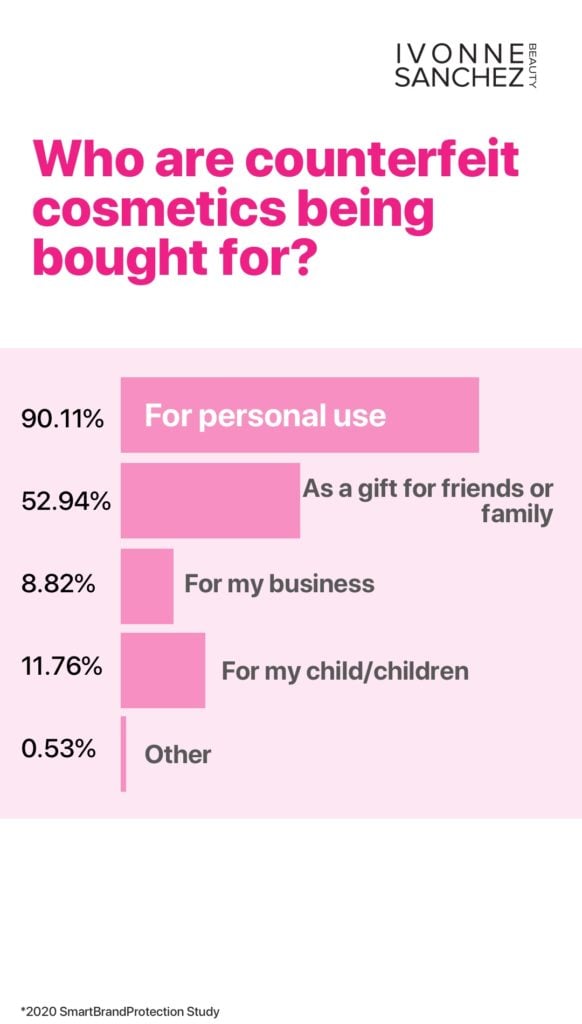
You’ve bought it from an unauthorized source: Buying a product from a source outside of the usual supply chain could mean that the product is counterfeit, containing questionable ingredients, uncertain ingredient concentrations, unvalidated expiry dates. Worse, the product may have been shipped in conditions that have compromised the effectiveness of the product. Think trucks that are too hot, left to sit in the sun.
You’ve bought this product from a massive online marketplace like Amazon, Facebook Marketplace, or some random account without any history on Kijiji.
Buying branded skincare or cosmetics at bargain basement prices or from non-traditional channels is taking a huge risk. There is very little control over the authenticity or safety of the product when you buy from non-sanctioned outlets. Is your retailer authorized? Call or consult with the manufacturer’s website to find out.
The product is new, or new to the Canadian Market
At IVONNE we have undertaken the laborious task to file all of our foreign products with Health Canada. In several instances when we have asked our suppliers for details they were caught off guard, were unaware of the requirements, or told us “we don’t need to worry about it”, or “we’ve never had a problem selling to Canada”, but that’s not the answer we’re looking for.
The internet gives us access to buy products from allover the world, but that doesn’t mean that we should. Buying a product from another country always comes with the risk that it hasn’t been qualified for use anywhere, that includes Canada. So doing a little digging will prevent you from running into a scary product that could be too potent.
Is It White-Label, or Private Label? Who Knows!
It has become increasingly popular for small beauty businesses to setup their own skincare or cosmetic brand with their logo. For a small investment of just $5,000 to $50,000 there are labs around the world that will ship custom branded products. More than ever there is a jungle of different brands all offering their own version of beauty.
Your Cosmetic Is Making Therapeutic Claims Without a DIN
If your skincare or cosmetic is claiming to have therapeutic benefits but doesn’t have a DIN, then chances are it hasn’t been reviewed in a Notification process. Health Canada closely adheres to the Ad Standards and Guidelines for the Nonprescription and Cosmetic Industry Regarding Non-therapeutic Advertising and Labelling Claims.
Who Can You Trust?
The more information – the better. When researching a cosmetic or skincare brand be sure to check out the manufacturers website, make sure that retailer is authorized as a provider, and ask them for evidence that the product has been approved for use and sale in Canada. Look for confirmation that the manufacturer and the provider have a good relationship (training sessions, social engagement etc). Avoid bargains and retailers that are questionable, and be sure to qualify any skincare routine with your dermatologist.
Don’t forget to read reviews and visit the Instagram feed. Many clients pose questions or feedback in online store platforms or Instagram posts. Are these questions and concerns being answered? Do the answers make sense?
Are Your Products Safe?